|
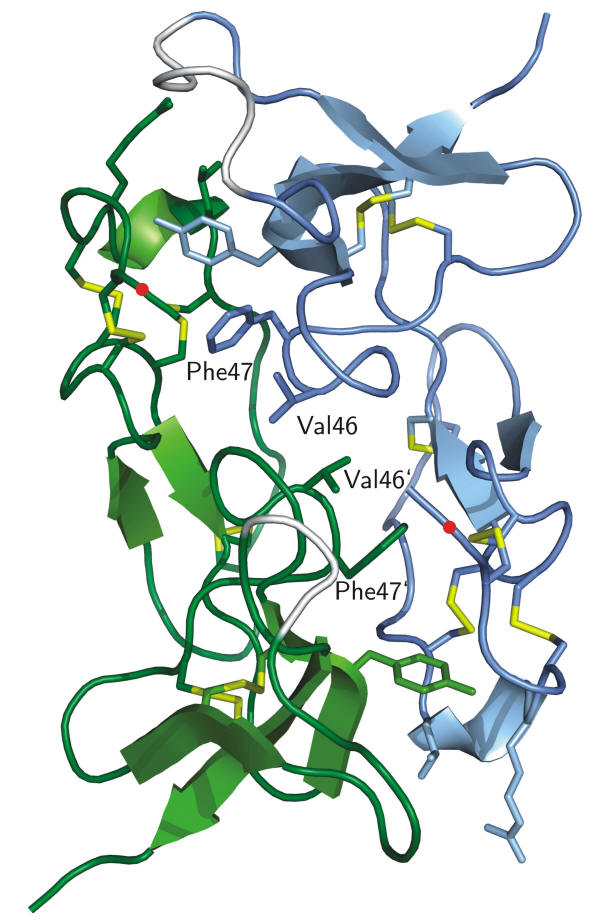
The 3D structure of recombinant SIBD-1 discovered in the hemocytes of the Central American hunting spider Cupiennius salei was determined by X-ray diffraction at 1.4 Å.
In the rSIBD-1 dimer chain A is coloured in blue, chain B is in green, and the six disulfide bridges are in yellow. Each monomer contains five b-strands and one a-helix. The hydrophobic dipeptides Val 46 and Phe 47 as part of the dimer interface are shown as sticks and labelled. The site of glycosylation at Thr 2 in native SIBD-1 is marked by a red dot. SIBD-1 seems to play an important role in the spiders immune system.
This work was carried out in the groups of Prof. Dr. Johann Schaller and PD Dr. Stefan Schürch, in the group of Prof. Dr. Wolfgang Nentwig / Dr. Lucia Kuhn-Nentwig at the Institute of Ecology and Evolution (University of Bern), and in the group of Prof. Dr. Ulrich Baumann at the Institute of Biochemistry (Cologne University, Germany).
References:
-
C. Trachsel, C. Widmer, U. Kämpfer, C. Bühr, T. Baumann, L. Kuhn-Nentwig, S. Schürch, J. Schaller, U. Baumann;
"Structural and biochemical characterization of native and recombinant single insulin-like growth factor-binding domain protein (SIBD-1) from the Central American hunting spider Cupiennius salei (Ctenidae)"
Proteins Struct. Funct. Bioinform., 80, 2323-2329, (2012);
doi:10.1002/prot.24119.
-
L. Kuhn-Nentwig, C. R. Largiadèr, K. Streitberger, S. Chandru, T. Baumann, U. Kämpfer, J. Schaller, S. Schürch, W. Nentwig;
"Purification, cDNA structure and biological significance of a single insulin-like growth factor-binding domain protein (SIBD-1) identified in the hemocytes of the spider Cupiennius salei"
Insect. Biochem. Mol. Biol., 41, 891-901, (2011);
doi:10.1016/j.ibmb.2011.08.003.
|